It is fundamentally different from the conventional periodic table in organizing entities by charge and consequently in showing many elements multiple times because of the multiple charges or valence states taken by those elements. From the website: "The Earth Scientist's Periodic Table of the Elements and Their Ions is a new periodic table designed to contextualize trends in geochemistry, mineralogy, aqueous chemistry, and other natural sciences. I have to believe that the CAST will include an online periodic table, so I have cobbled together the equations and constants from CAST that I think will apply to Chemistry, and combined it here with an up-to-date periodic table in a two-sided resource sheet that my students will be using in class.Īn Earth Scientist's Periodic Table, this is available in number of sizes on the linked page. Also, the California Department of Education recently published a set of equations and constants as a resource for our new CAST (California Science Test). I recently decided that the old CA State table we used for years needed to go away. When you are all done creating your masterpiece, you can download it as a pdf file. You can also customize font size and add a logo. Atomic mass, electron configuration, density, boiling point, freezing point - all can be included by simply checking a box. This is online software that allows an amazing amount of customization, from color schemes to the data that you want displayed. This is the second "Build Your Own Table" website to which I have linked. If the file does not reside on my server, you will be directed to a page where the table CAN be downloaded. Only seven of these tables are my creation all rights are retained by the original authors of the others.Ĭlick on the element symbol to download the PDF file. The quality of the results will depend in large part on the quality of your printer. All of the tables will print on a single 8½ x 11 page, though some may require color to retain their appearance. The following descriptions include a sample element from each periodic table.
The program can be downloaded from the Adobe Website. To view and print these files, you will need to install the free Adobe Acrobat Reader program on your computer.
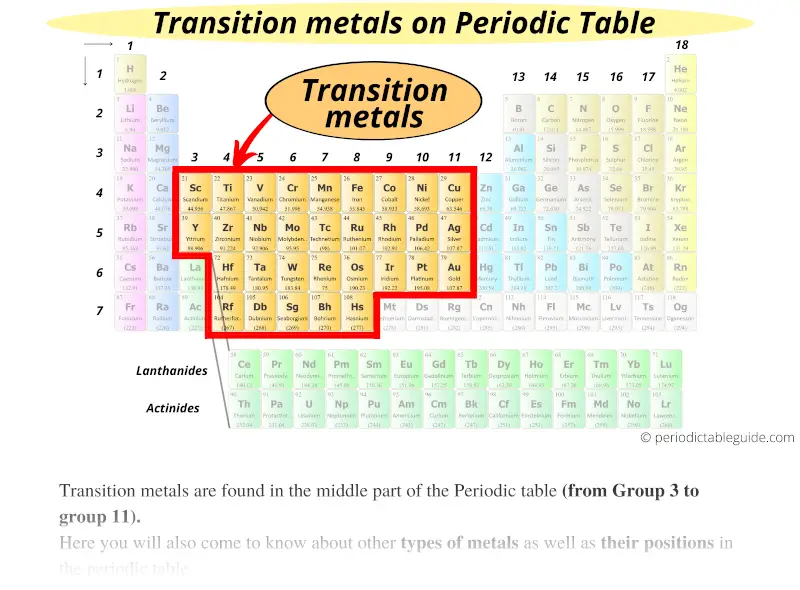
The positive oxidation state means the transition metals typically form ionic or partially ionic compounds. Copper may have a 1+ or 2+ oxidation state. For example, iron commonly carries a 3+ or 2+ oxidation state. Because the subshell is not filled, atoms of the transition metals have positive oxidation states and also display more than one oxidation state. Their d orbitals become progressively filled as you move from left to right across the periodic table.By extension, these elements also have high boiling points. The exception is mercury, which is a liquid at room temperature. The transition metals, as a group, have high melting points.Most transition metals are grayish or white (like iron or silver), but gold and copper have colors not seen in any other element on the periodic table. Transition metals look shiny and metallic.The transition metals are malleable (easily hammered into shape or bent).They are excellent conductors of heat and electricity.

